Volume 9, Issue 1 (Continuously Updated- In press 2026)
Func Disabil J 2026, 9(1): 0-0 |
Back to browse issues page
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
Nazari Z, Vassaghi Gharamaleki B, Babazadeh S. Spirometric Indices in Semi-professional Female Athletes With Stress Urinary Incontinence: A Case-Control study. Func Disabil J 2026; 9 (1)
URL: http://fdj.iums.ac.ir/article-1-338-en.html
URL: http://fdj.iums.ac.ir/article-1-338-en.html
1- Department of Physiotherapy, School of Rehabilitation Sciences, Iran University of Medical Sciences, Tehran, Iran.
2- Department of Rehabilitation Basic Sciences, Rehabilitation Research Center, School of Rehabilitation Sciences, Iran University of Medical Sciences, Tehran, Iran. ,bvasaghi@gmail.com
3- Department of Physical Therapy, School of Rehabilitation, Arak University of Medical Sciences, Arak, Iran.
2- Department of Rehabilitation Basic Sciences, Rehabilitation Research Center, School of Rehabilitation Sciences, Iran University of Medical Sciences, Tehran, Iran. ,
3- Department of Physical Therapy, School of Rehabilitation, Arak University of Medical Sciences, Arak, Iran.
Keywords: Stress urinary incontinence (SUI), Female athletes, Spirometry, Peak expiratory flow (PEF), Vital capacity (VC), Diaphragm, Pelvic floor muscles
Full-Text [PDF 859 kb]
(6 Downloads)
| Abstract (HTML) (61 Views)
Full-Text: (3 Views)
Introduction
Stress urinary incontinence (SUI) is the most prevalent type of urinary incontinence, defined as the involuntary leakage of urine during activities that increase intra-abdominal pressure, such as coughing, sneezing, running, and jumping [1-3]. Its overall prevalence in women is high, reaching approximately 34% in Iranian women, and is even more concerning among female athletes who are otherwise considered healthy and physically strong [4]. Systematic reviews estimate that nearly 25–30% of female athletes experience urinary incontinence, with the majority presenting as SUI. High-impact sports, such as volleyball, basketball, aerobics, and gymnastics report the greatest burden, with volleyball players showing prevalence rates as high as 75% [5, 6].
Although SUI is traditionally associated with multiparity and pelvic floor trauma, its presence in nulliparous athletes underscores distinct pathophysiological mechanisms. In athletes, repetitive mechanical loading, elevated intra-abdominal pressure, and altered breathing mechanics are central contributors [7-9]. The pelvic floor, diaphragm, and abdominal muscles function synergistically to maintain continence while regulating respiration [10]. During inspiration, the diaphragm descends while the pelvic floor eccentrically lengthens to accommodate abdominal displacement; during expiration, both structures contract synergistically to stabilize the trunk and maintain continence [11]. Disruption of this synergy, as occurs with SUI, may impair both continence and ventilatory performance [12].
Emerging evidence suggests that SUI is associated with respiratory impairments. Studies on non-athletic women with SUI have reported reduced respiratory muscle strength, particularly expiratory pressures, and decreased ventilatory volumes [13, 14]. Several studies have demonstrated that women with SUI show significantly reduced respiratory muscle strength, particularly in expiratory pressures, compared with healthy controls [15]. Research also indicates that interventions targeting the pelvic floor can improve diaphragm excursion, rib cage movement, and ventilatory performance [16]. Moreover, contractions of the pelvic floor appear to influence diaphragmatic motion and spirometric outputs, reinforcing the concept that impaired diaphragm–pelvic floor coordination may underlie both respiratory dysfunction and urinary incontinence [14].
However, data on semi-professional female athletes remain scarce. Despite the assumption of superior muscular conditioning, these athletes are not immune to SUI and may even be at increased risk due to repetitive intra-abdominal loading [17-19]. No study has comprehensively compared spirometric indices between athletes with SUI and their healthy peers. Assessing these indices may provide insight into the functional consequences of impaired diaphragm–pelvic floor synergy in this unique population.
Therefore, the present study aimed to evaluate and compare spirometric parameters in semi-professional female athletes with and without SUI. We hypothesized that athletes with SUI would demonstrate reduced inspiratory capacities, expiratory flows, and altered ventilatory timing compared to controls, reflecting dysfunction in core muscle synergy.
Materials and Methods
Study design
This cross-sectional case–control study was conducted on semi-professional female athletes in Tehran. The study compared spirometric indices between athletes with clinically confirmed SUI and healthy athletes without incontinence.
Sample size determination
Sample size was determined using G*Power software, version 3.1.9.4, assuming a medium effect size, a significance level of 0.05, and a statistical power of 80%. The calculation yielded a minimum of 18 participants per group. To account for potential dropouts, 20 participants were recruited for each group, yielding a total of 40 athletes.
Eligibility criteria
Participants were women aged 18–38 years, actively training in high-impact sports (volleyball, basketball, gymnastics, and aerobics) for at least 150 minutes per week during the past six months. The case group included athletes diagnosed with SUI using the validated Persian version of the international consultation on incontinence questionnaire–urinary incontinence short form (ICIQ-UI-SF). Exclusion criteria were a history of childbirth, pelvic surgery, chronic respiratory or cardiovascular disease, smoking, neurological or metabolic disorders, and recent musculoskeletal injuries. Controls met identical criteria except for the absence of urinary incontinence.
Information collection method
Baseline demographic and anthropometric data, including age, height, weight, and body mass index (BMI) were recorded. Sport type and training history were documented. The ICIQ-UI-SF was administered to confirm group allocation. Spirometry was performed in accordance with the American Thoracic Society/European respiratory society guidelines, measuring expiratory reserve volume (ERV), inspiratory reserve volume (IRV), tidal volume (TV), vital capacity (VC), inspiratory vital capacity (IVC), inspiratory capacity (IC), forced expiratory volume in one second (FEV1), forced vital capacity (FVC), FEV1/FVC ratio, peak expiratory flow (PEF), inspiratory time (TI), and expiratory time (TE). All measurements were performed in a sitting position using a calibrated KALAMED PC-Spirometer (Model KSP-1000, Serial No: ST-14070062; Polymed Group, Suisse), with at least three reproducible attempts obtained per participant.
Statistical analysis
Data were analyzed using SPSS software, version 18. Normality of data distribution was tested using the Shapiro–Wilk test. Continuous variables were expressed as Mean±SD and compared between groups using independent t-tests. Categorical variables, including sport type, were compared using chi-square tests. Statistical significance was set at P<0.05.
Results
Patients’ characteristics
A total of 40 semi-professional female athletes were evaluated, comprising 20 athletes diagnosed with SUI and 20 athletes without SUI (this term was used instead of “control group” to avoid implying complete health status, as the absence of other conditions was not confirmed). The athletes participated in four sport disciplines; volleyball (11), basketball (11), gymnastics (9), and aerobics (9). The mean age in the SUI group was 28.70±5.77 years, while the group without SUI averaged 27.60±5.61 years; this difference was not statistically significant (P=0.544). Similarly, mean height and weight were not significantly different between the groups (P=0.593 and P=0.156, respectively). The two groups were therefore matched with respect to age and anthropometric characteristics.
The only anthropometric parameter that showed a significant difference was BMI, which was lower in the SUI group (21.09±1.79 kg/m²) compared to controls (22.22±1.43 kg/m², P=0.0305). This finding suggests that SUI in athletes cannot simply be attributed to elevated BMI, as is commonly observed in non-athletic populations. The detailed demographic and anthropometric results are presented in Table 1.

Sport distribution
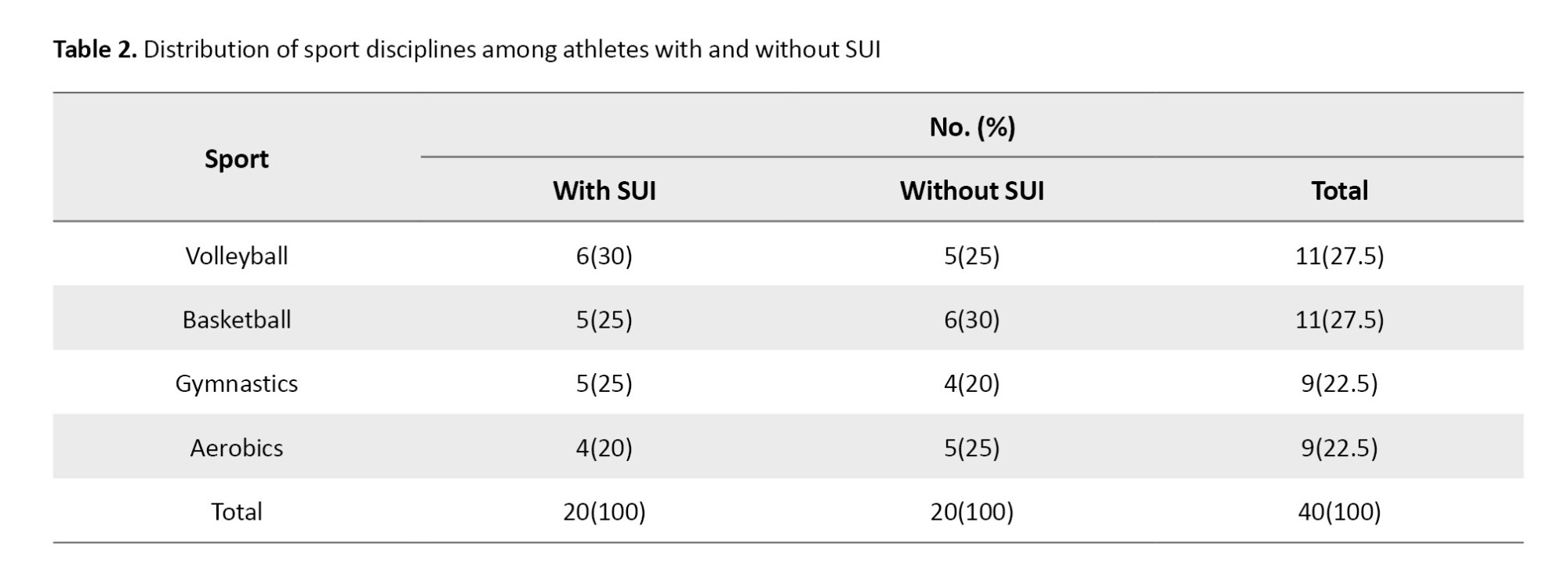
The distribution of participants across the four high-impact sports (volleyball, basketball, gymnastics, and aerobics) was balanced between the two groups. For example, volleyball represented 30% of the SUI group and 25% of the controls, while basketball was reported in 25% and 30%, respectively. Gymnastics and aerobics also showed nearly equal representation between groups. Statistical comparison confirmed no significant difference in the distribution of sport type (χ²=2.54, P=0.469). This indicates that the observed differences in spirometric performance cannot be explained by the type of sport type practiced, strengthening the inference that the differences are linked to continence status (Table 2).
Spirometric indices
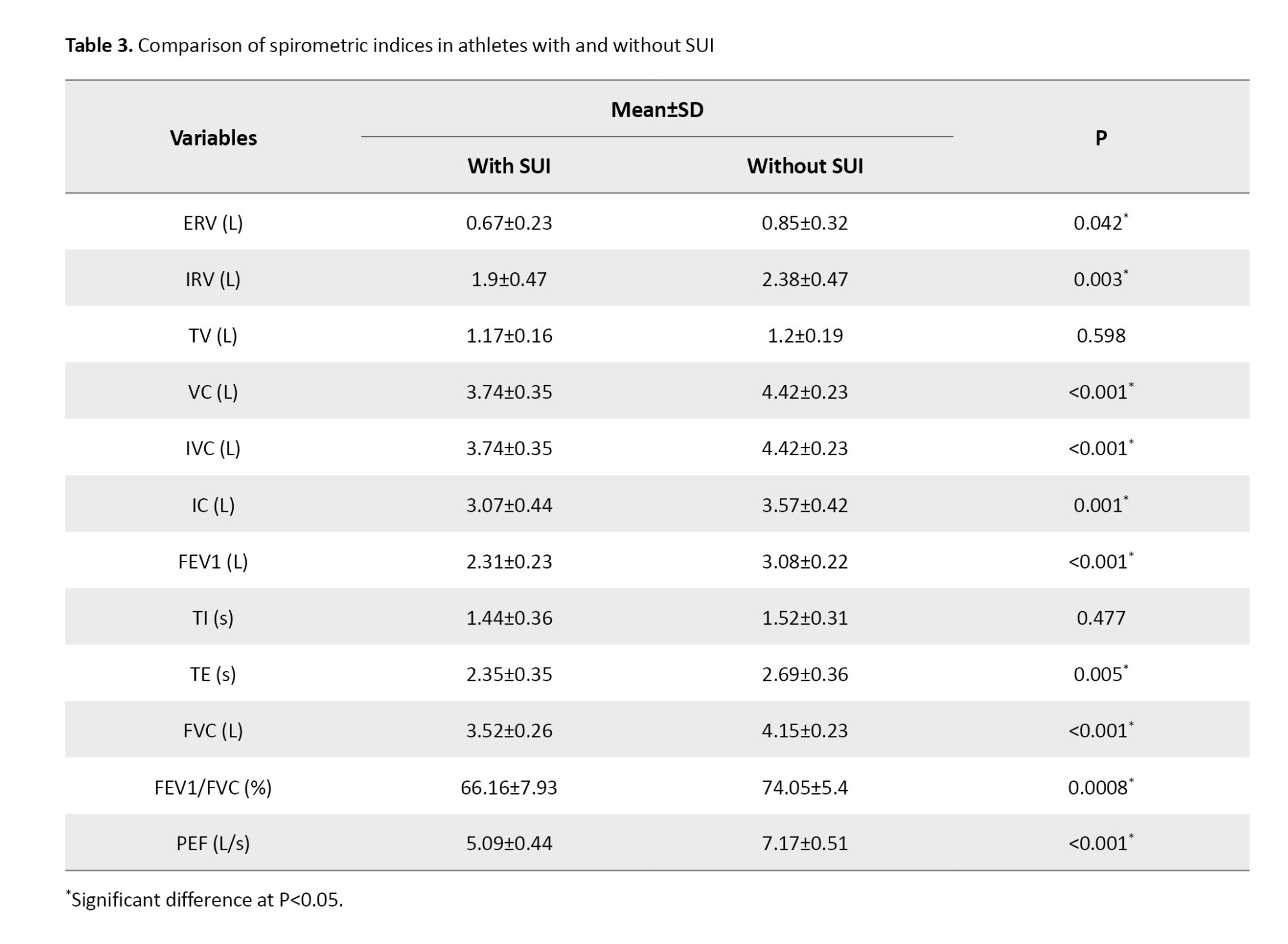
Significant between-group differences were observed in multiple spirometric indices. FEV1 was substantially lower in athletes with SUI (2.31±0.23 L) compared with controls (3.08±0.22 L, P<0.001). Similarly, FVC was reduced in the SUI group (3.52±0.26 L) versus controls (4.15±0.23 L, P<0.001). The FEV1/FVC ratio, a sensitive marker of expiratory flow limitation, was also markedly reduced in SUI athletes (66.16±7.93%) compared with controls (74.05±5.4%, P=0.0008).
PEF, which reflects the maximal strength and speed of expiratory effort, was significantly impaired in the SUI group (5.09±0.44 L/s vs 7.17±0.51 L/s, P<0.001).
Beyond these primary measures, capacities reflecting inspiratory and expiratory reserves were also reduced. VC, IVC, IC, IRV, and ERV were all significantly lower in the SUI group (P≤0.005). For example, mean IRV was 1.90±0.47 L in the SUI group compared with 2.38±0.47 L in controls (P=0.003). Similarly, ERV was reduced to 0.67±0.23 L in the SUI group versus 0.85±0.32 L in controls (P=0.042).
By contrast, TV and TI did not differ significantly, suggesting that resting breathing patterns remain preserved. However, TE was significantly shorter in SUI athletes (2.35±0.35 s vs 2.69±0.36 s, P=0.005), indicating a limitation in the duration and efficiency of exhalation. Detailed data are shown in Table 3, while the main differences in FVC, FEV1, FEV1/FVC, and PEF are illustrated in Figure 1.
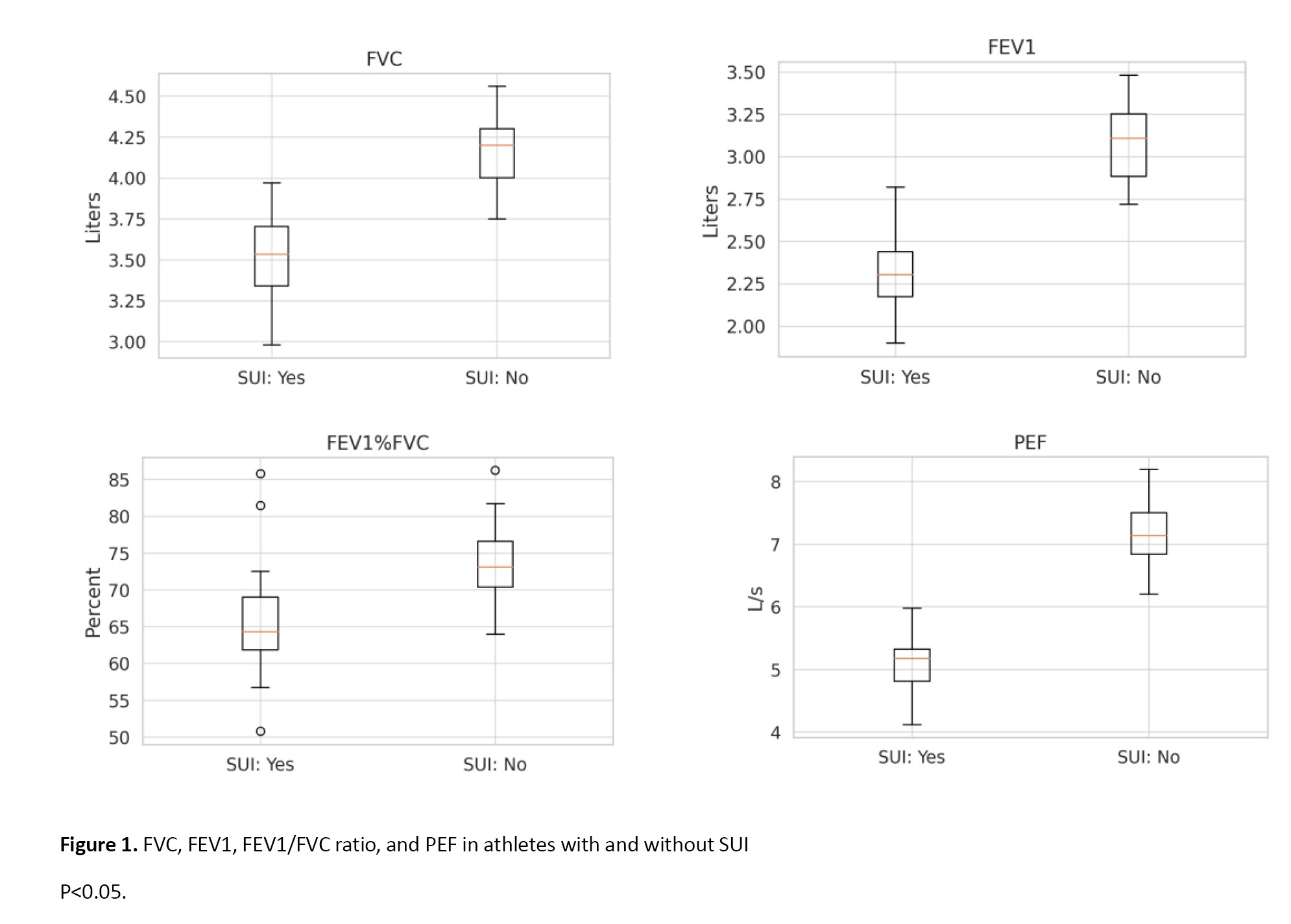
Flow-volume curves
Qualitative assessment of flow–volume loops demonstrated distinct alterations in athletes with SUI. Specifically, the PEF was blunted, and the ascending slope of the expiratory limb was less steep compared to controls. This suggests weaker initial expiratory effort, likely related to reduced abdominal and pelvic floor muscle coordination. In addition, the overall area under the expiratory curve appeared reduced, consistent with diminished FVC values.
Representative curves are shown in Figure 2, where the reduced peak flow and flattened slope are evident in the SUI group.
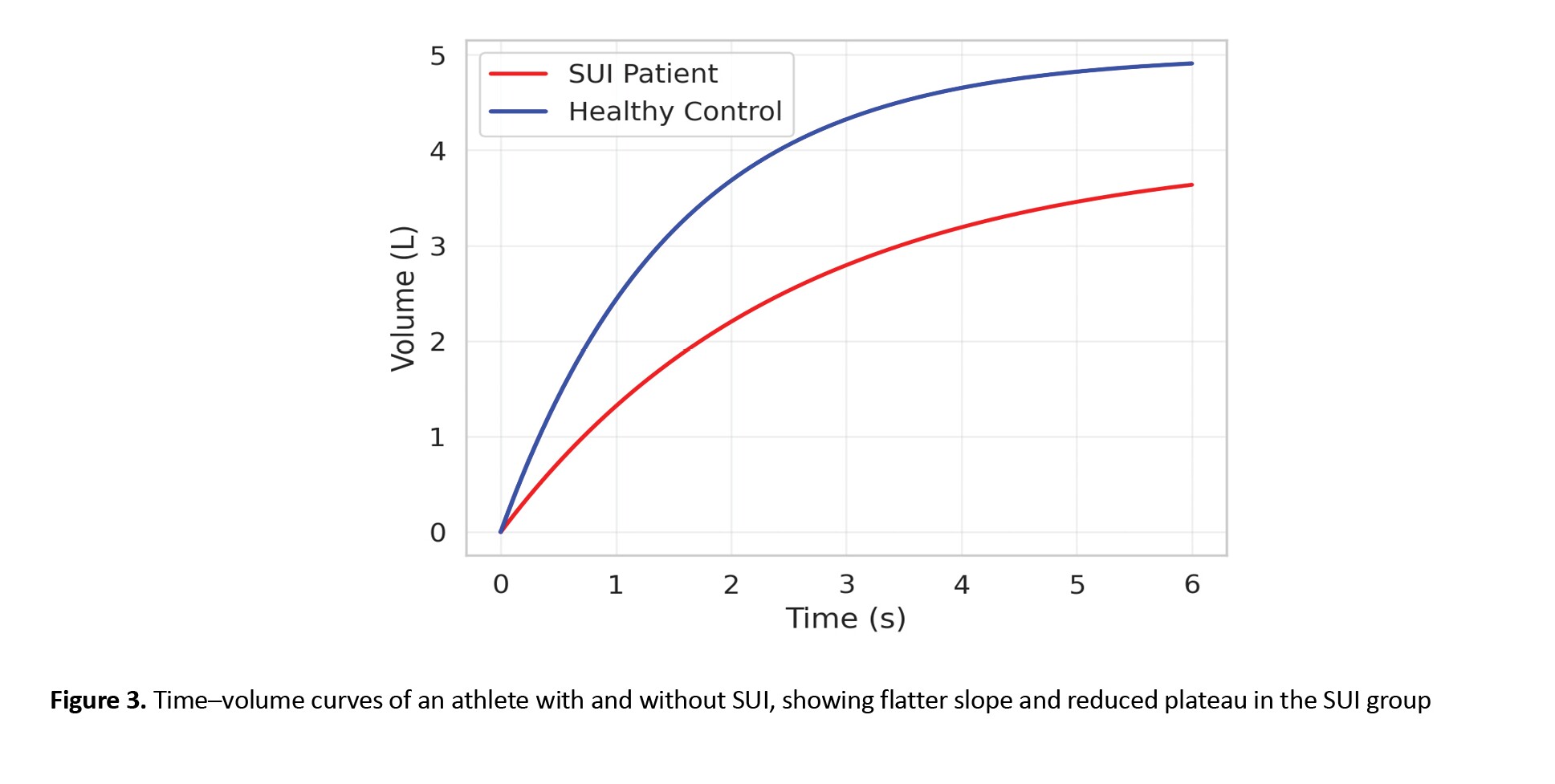
Time–volume curves
Time–volume curve analysis also revealed notable differences between groups. Athletes with SUI demonstrated slower rises in volume during forced expirations, reflecting lower FEV1 values, and required more time to approach final lung volumes. The slope of the curve during the first second was consistently flatter in SUI athletes, confirming the quantitative reductions in FEV1.
Additionally, the plateau phase of the time–volume curves was lower in the SUI group, consistent with reduced FVC. These graphical findings further support the quantitative data indicating compromised expiratory performance. Examples of time–volume curves for both groups are displayed in Figure 3.
Discussion
The primary aim of this study was to compare spirometric indices in semi-professional female athletes with SUI versus healthy controls. Consistent and clinically meaningful reductions in volume and flow metrics among athletes with SUI include lower VC, IC, ERV, IVC, IRV, as well as reductions in FVC, FEV1, FEV1/FVC, and PEF, while TV and TI did not differ. This pattern indicates that SUI in athletes is not only associated with reduced vital capacities but also with expiratory flow limitation and altered ventilatory timing, pointing toward dysfunction in respiratory muscle performance, particularly the diaphragm as the primary inspiratory muscle, and disturbed synergy across the diaphragm, abdominal wall, and pelvic floor complex.
The absence of between-group differences in TV and TI suggests that quiet tidal breathing mechanics remain preserved, whereas the significant reductions in IRV and IC support an inspiratory constraint that is compatible with suboptimal diaphragmatic excursion or efficiency. The larger decrement in IRV than ERV is physiologically consistent with the diaphragm’s dominant role in generating inspiratory reserve; increased abdominal wall stiffness or suboptimal diaphragmatic length–tension could further accentuate this effect. The ERV deficit, in turn, likely indexes reduced contribution of the abdominal wall during forced expiration, again highlighting the interdependence between abdominal muscle function and pelvic floor control in athletes with SUI.
Our findings align with reports that women with incontinence exhibit diminished respiratory muscle strength, especially on the expiratory side, and poorer physical function [13]. The significantly lower PEF observed in SUI athletes mirrors the reduction in maximal expiratory performance previously described, supporting the notion that impaired abdominal and pelvic floor recruitment limits the generation of rapid, forceful expiratory flow [13]. In the context of sport, where repetitive surges in intra-abdominal pressure are common, such deficits plausibly exacerbate continence challenges.
Experimental work demonstrates that modulating pelvic floor function can alter ventilatory mechanics, reinforcing a bidirectional diaphragm–pelvic floor relationship. Laboratory studies have shown that voluntary pelvic floor contractions influence diaphragmatic motion and dynamic ventilation outputs, including changes in FEV1 and maximum ventilatory volume [14, 20]. Clinical trials extend this link: targeted pelvic floor electrical stimulation improved diaphragm excursion and upper rib cage movement during both tidal and forceful breathing, together with gains in pelvic floor strength [21]. Exercise-based interventions that combine breathing training with pelvic floor–focused activity have reduced SUI severity and improved pelvic floor performance and quality of life [22]. Taken together with our results, these data suggest that restoring coordinated activation across pelvic floor and abdominal compartments may secondarily normalize spirometric performance, particularly IRV and IC on the inspiratory side and PEF and FEV1 on the expiratory side, by improving diaphragmatic mechanics and abdominal pressurization [14, 19-22].
The reduced VC and IVC in our athletes with SUI are expected consequences of the concomitant IRV and ERV decrements and are consistent with the basic physiological model in which pelvic floor and abdominal muscles co-contract with the diaphragm during expiration to stabilize the trunk and regulate intra-abdominal pressure [23]. When this synergy is perturbed, the diaphragm may operate at a less favorable length or excursion, diminishing inspiratory reserve and overall vital capacity [23]. The significantly lower IC in the SUI group further supports this interpretation; while low IC can be seen in airway disease, the absence of respiratory pathology and the athletic status of our controls make a neuromuscular or synergy explanation more plausible here.
Functionally, the reduced FEV1, FVC, and FEV1/FVC ratio in SUI athletes, despite no clinical evidence of obstructive disease, likely reflects submaximal coordinated expiratory effort rather than intrinsic airflow obstruction. Prior experimental work has shown pelvic floor activity to modulate key spirometric outputs without necessarily changing static lung mechanics [14, 20]. The shorter TE in our SUI cohort complements this picture, indicating earlier cessation of forced exhalation, consistent with limited capacity to sustain high expiratory flows. Graphical analyses support the quantitative results: a blunted peak flow and a shallower early expiratory slope on flow–volume loops, together with a flatter rise in the first second and a lower plateau on time–volume curves, are typical of impaired expiratory muscle recruitment and reduced FEV1 and PEF [24].
Epidemiologically, our cohort reflects the broader pattern of elevated SUI burden in high-impact sports, where repetitive jumps, landings, and trunk bracing demand precise diaphragm–pelvic floor timing to maintain continence [18]. The present data add a mechanistic layer, showing that even in well-trained athletes, SUI is accompanied by measurable impairments on routine spirometry. Clinically, this suggests that selected spirometric markers, especially FEV1, FVC, PEF, and FEV1/FVC, can serve as accessible adjuncts for screening athletes with SUI and tracking responses to integrated respiratory–pelvic floor rehabilitation [13, 14, 20-23].
Overall, our results converge with prior evidence from laboratory, clinical, and interventional contexts: SUI is associated with altered diaphragm–pelvic floor synergy, observable as reduced inspiratory reserves and compromised expiratory flows. In athletes, where intra-abdominal pressure dynamics are repeatedly stressed, these alterations may be both more apparent and more consequential for performance and continence.
Conclusion
Semi-professional female athletes with SUI exhibit significant impairments in both inspiratory and expiratory spirometric indices compared with their healthy peers. These impairments include reduced vital capacities, diminished expiratory flows, and shortened expiratory time, suggesting dysfunctional coordination of the diaphragm, abdominal muscles, and pelvic floor. Despite their regular engagement in high-intensity sports, athletes with SUI demonstrate measurable limitations in ventilatory performance, highlighting the central role of core muscle synergy in both respiration and continence. The use of standard spirometry, particularly parameters, such as FEV1, FVC, PEF, and FEV1/FVC, may provide a valuable and accessible method for screening athletes at risk and for evaluating responses to rehabilitation programs. Targeted interventions that integrate pelvic floor and respiratory training could be essential components of management in this population.
Limitations
This cross-sectional design used limits causal inference regarding whether altered ventilatory mechanics contribute to SUI or result from it. SUI classification relied on a validated questionnaire rather than urodynamic testing, which could strengthen diagnostic precision. The sample size, while powered for primary comparisons, constrained finer subgroup analyses by sport or training load. We did not include direct measures of respiratory muscle strength or imaging of diaphragmatic excursion, which would help disentangle inspiratory versus expiratory contributions to the observed pattern. Finally, findings pertain to semi-professional athletes in selected high-impact sports and may not generalize to elite competitors, recreational athletes, or non-athletic women.
Ethical Considerations
Compliance with ethical guidelines
This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Iran University of Medical Sciences, Tehran, Iran (Code: IR.IUMS.REC.1403.525). As this was a retrospective study based on anonymized records, the requirement for informed consent was waived by the ethics committee.
Funding
This research did not receive any grant from funding agencies in the public, commercial, or non-profit sectors.
Authors' contributions
Conceptualization, data collection, data analysis, and drafting of the manuscript: Zahra Nazari; Study supervision, methodology, and critical revision of the manuscript: Behnoush Vassaghi Gharamaleki and Saeedeh Babazadeh.
Conflict of interest
The authors declared no conflict of interest.
References
Stress urinary incontinence (SUI) is the most prevalent type of urinary incontinence, defined as the involuntary leakage of urine during activities that increase intra-abdominal pressure, such as coughing, sneezing, running, and jumping [1-3]. Its overall prevalence in women is high, reaching approximately 34% in Iranian women, and is even more concerning among female athletes who are otherwise considered healthy and physically strong [4]. Systematic reviews estimate that nearly 25–30% of female athletes experience urinary incontinence, with the majority presenting as SUI. High-impact sports, such as volleyball, basketball, aerobics, and gymnastics report the greatest burden, with volleyball players showing prevalence rates as high as 75% [5, 6].
Although SUI is traditionally associated with multiparity and pelvic floor trauma, its presence in nulliparous athletes underscores distinct pathophysiological mechanisms. In athletes, repetitive mechanical loading, elevated intra-abdominal pressure, and altered breathing mechanics are central contributors [7-9]. The pelvic floor, diaphragm, and abdominal muscles function synergistically to maintain continence while regulating respiration [10]. During inspiration, the diaphragm descends while the pelvic floor eccentrically lengthens to accommodate abdominal displacement; during expiration, both structures contract synergistically to stabilize the trunk and maintain continence [11]. Disruption of this synergy, as occurs with SUI, may impair both continence and ventilatory performance [12].
Emerging evidence suggests that SUI is associated with respiratory impairments. Studies on non-athletic women with SUI have reported reduced respiratory muscle strength, particularly expiratory pressures, and decreased ventilatory volumes [13, 14]. Several studies have demonstrated that women with SUI show significantly reduced respiratory muscle strength, particularly in expiratory pressures, compared with healthy controls [15]. Research also indicates that interventions targeting the pelvic floor can improve diaphragm excursion, rib cage movement, and ventilatory performance [16]. Moreover, contractions of the pelvic floor appear to influence diaphragmatic motion and spirometric outputs, reinforcing the concept that impaired diaphragm–pelvic floor coordination may underlie both respiratory dysfunction and urinary incontinence [14].
However, data on semi-professional female athletes remain scarce. Despite the assumption of superior muscular conditioning, these athletes are not immune to SUI and may even be at increased risk due to repetitive intra-abdominal loading [17-19]. No study has comprehensively compared spirometric indices between athletes with SUI and their healthy peers. Assessing these indices may provide insight into the functional consequences of impaired diaphragm–pelvic floor synergy in this unique population.
Therefore, the present study aimed to evaluate and compare spirometric parameters in semi-professional female athletes with and without SUI. We hypothesized that athletes with SUI would demonstrate reduced inspiratory capacities, expiratory flows, and altered ventilatory timing compared to controls, reflecting dysfunction in core muscle synergy.
Materials and Methods
Study design
This cross-sectional case–control study was conducted on semi-professional female athletes in Tehran. The study compared spirometric indices between athletes with clinically confirmed SUI and healthy athletes without incontinence.
Sample size determination
Sample size was determined using G*Power software, version 3.1.9.4, assuming a medium effect size, a significance level of 0.05, and a statistical power of 80%. The calculation yielded a minimum of 18 participants per group. To account for potential dropouts, 20 participants were recruited for each group, yielding a total of 40 athletes.
Eligibility criteria
Participants were women aged 18–38 years, actively training in high-impact sports (volleyball, basketball, gymnastics, and aerobics) for at least 150 minutes per week during the past six months. The case group included athletes diagnosed with SUI using the validated Persian version of the international consultation on incontinence questionnaire–urinary incontinence short form (ICIQ-UI-SF). Exclusion criteria were a history of childbirth, pelvic surgery, chronic respiratory or cardiovascular disease, smoking, neurological or metabolic disorders, and recent musculoskeletal injuries. Controls met identical criteria except for the absence of urinary incontinence.
Information collection method
Baseline demographic and anthropometric data, including age, height, weight, and body mass index (BMI) were recorded. Sport type and training history were documented. The ICIQ-UI-SF was administered to confirm group allocation. Spirometry was performed in accordance with the American Thoracic Society/European respiratory society guidelines, measuring expiratory reserve volume (ERV), inspiratory reserve volume (IRV), tidal volume (TV), vital capacity (VC), inspiratory vital capacity (IVC), inspiratory capacity (IC), forced expiratory volume in one second (FEV1), forced vital capacity (FVC), FEV1/FVC ratio, peak expiratory flow (PEF), inspiratory time (TI), and expiratory time (TE). All measurements were performed in a sitting position using a calibrated KALAMED PC-Spirometer (Model KSP-1000, Serial No: ST-14070062; Polymed Group, Suisse), with at least three reproducible attempts obtained per participant.
Statistical analysis
Data were analyzed using SPSS software, version 18. Normality of data distribution was tested using the Shapiro–Wilk test. Continuous variables were expressed as Mean±SD and compared between groups using independent t-tests. Categorical variables, including sport type, were compared using chi-square tests. Statistical significance was set at P<0.05.
Results
Patients’ characteristics
A total of 40 semi-professional female athletes were evaluated, comprising 20 athletes diagnosed with SUI and 20 athletes without SUI (this term was used instead of “control group” to avoid implying complete health status, as the absence of other conditions was not confirmed). The athletes participated in four sport disciplines; volleyball (11), basketball (11), gymnastics (9), and aerobics (9). The mean age in the SUI group was 28.70±5.77 years, while the group without SUI averaged 27.60±5.61 years; this difference was not statistically significant (P=0.544). Similarly, mean height and weight were not significantly different between the groups (P=0.593 and P=0.156, respectively). The two groups were therefore matched with respect to age and anthropometric characteristics.
The only anthropometric parameter that showed a significant difference was BMI, which was lower in the SUI group (21.09±1.79 kg/m²) compared to controls (22.22±1.43 kg/m², P=0.0305). This finding suggests that SUI in athletes cannot simply be attributed to elevated BMI, as is commonly observed in non-athletic populations. The detailed demographic and anthropometric results are presented in Table 1.

Sport distribution
The distribution of participants across the four high-impact sports (volleyball, basketball, gymnastics, and aerobics) was balanced between the two groups. For example, volleyball represented 30% of the SUI group and 25% of the controls, while basketball was reported in 25% and 30%, respectively. Gymnastics and aerobics also showed nearly equal representation between groups. Statistical comparison confirmed no significant difference in the distribution of sport type (χ²=2.54, P=0.469). This indicates that the observed differences in spirometric performance cannot be explained by the type of sport type practiced, strengthening the inference that the differences are linked to continence status (Table 2).

Spirometric indices
Significant between-group differences were observed in multiple spirometric indices. FEV1 was substantially lower in athletes with SUI (2.31±0.23 L) compared with controls (3.08±0.22 L, P<0.001). Similarly, FVC was reduced in the SUI group (3.52±0.26 L) versus controls (4.15±0.23 L, P<0.001). The FEV1/FVC ratio, a sensitive marker of expiratory flow limitation, was also markedly reduced in SUI athletes (66.16±7.93%) compared with controls (74.05±5.4%, P=0.0008).
PEF, which reflects the maximal strength and speed of expiratory effort, was significantly impaired in the SUI group (5.09±0.44 L/s vs 7.17±0.51 L/s, P<0.001).
Beyond these primary measures, capacities reflecting inspiratory and expiratory reserves were also reduced. VC, IVC, IC, IRV, and ERV were all significantly lower in the SUI group (P≤0.005). For example, mean IRV was 1.90±0.47 L in the SUI group compared with 2.38±0.47 L in controls (P=0.003). Similarly, ERV was reduced to 0.67±0.23 L in the SUI group versus 0.85±0.32 L in controls (P=0.042).
By contrast, TV and TI did not differ significantly, suggesting that resting breathing patterns remain preserved. However, TE was significantly shorter in SUI athletes (2.35±0.35 s vs 2.69±0.36 s, P=0.005), indicating a limitation in the duration and efficiency of exhalation. Detailed data are shown in Table 3, while the main differences in FVC, FEV1, FEV1/FVC, and PEF are illustrated in Figure 1.


Flow-volume curves
Qualitative assessment of flow–volume loops demonstrated distinct alterations in athletes with SUI. Specifically, the PEF was blunted, and the ascending slope of the expiratory limb was less steep compared to controls. This suggests weaker initial expiratory effort, likely related to reduced abdominal and pelvic floor muscle coordination. In addition, the overall area under the expiratory curve appeared reduced, consistent with diminished FVC values.
Representative curves are shown in Figure 2, where the reduced peak flow and flattened slope are evident in the SUI group.

Time–volume curves
Time–volume curve analysis also revealed notable differences between groups. Athletes with SUI demonstrated slower rises in volume during forced expirations, reflecting lower FEV1 values, and required more time to approach final lung volumes. The slope of the curve during the first second was consistently flatter in SUI athletes, confirming the quantitative reductions in FEV1.
Additionally, the plateau phase of the time–volume curves was lower in the SUI group, consistent with reduced FVC. These graphical findings further support the quantitative data indicating compromised expiratory performance. Examples of time–volume curves for both groups are displayed in Figure 3.

Discussion
The primary aim of this study was to compare spirometric indices in semi-professional female athletes with SUI versus healthy controls. Consistent and clinically meaningful reductions in volume and flow metrics among athletes with SUI include lower VC, IC, ERV, IVC, IRV, as well as reductions in FVC, FEV1, FEV1/FVC, and PEF, while TV and TI did not differ. This pattern indicates that SUI in athletes is not only associated with reduced vital capacities but also with expiratory flow limitation and altered ventilatory timing, pointing toward dysfunction in respiratory muscle performance, particularly the diaphragm as the primary inspiratory muscle, and disturbed synergy across the diaphragm, abdominal wall, and pelvic floor complex.
The absence of between-group differences in TV and TI suggests that quiet tidal breathing mechanics remain preserved, whereas the significant reductions in IRV and IC support an inspiratory constraint that is compatible with suboptimal diaphragmatic excursion or efficiency. The larger decrement in IRV than ERV is physiologically consistent with the diaphragm’s dominant role in generating inspiratory reserve; increased abdominal wall stiffness or suboptimal diaphragmatic length–tension could further accentuate this effect. The ERV deficit, in turn, likely indexes reduced contribution of the abdominal wall during forced expiration, again highlighting the interdependence between abdominal muscle function and pelvic floor control in athletes with SUI.
Our findings align with reports that women with incontinence exhibit diminished respiratory muscle strength, especially on the expiratory side, and poorer physical function [13]. The significantly lower PEF observed in SUI athletes mirrors the reduction in maximal expiratory performance previously described, supporting the notion that impaired abdominal and pelvic floor recruitment limits the generation of rapid, forceful expiratory flow [13]. In the context of sport, where repetitive surges in intra-abdominal pressure are common, such deficits plausibly exacerbate continence challenges.
Experimental work demonstrates that modulating pelvic floor function can alter ventilatory mechanics, reinforcing a bidirectional diaphragm–pelvic floor relationship. Laboratory studies have shown that voluntary pelvic floor contractions influence diaphragmatic motion and dynamic ventilation outputs, including changes in FEV1 and maximum ventilatory volume [14, 20]. Clinical trials extend this link: targeted pelvic floor electrical stimulation improved diaphragm excursion and upper rib cage movement during both tidal and forceful breathing, together with gains in pelvic floor strength [21]. Exercise-based interventions that combine breathing training with pelvic floor–focused activity have reduced SUI severity and improved pelvic floor performance and quality of life [22]. Taken together with our results, these data suggest that restoring coordinated activation across pelvic floor and abdominal compartments may secondarily normalize spirometric performance, particularly IRV and IC on the inspiratory side and PEF and FEV1 on the expiratory side, by improving diaphragmatic mechanics and abdominal pressurization [14, 19-22].
The reduced VC and IVC in our athletes with SUI are expected consequences of the concomitant IRV and ERV decrements and are consistent with the basic physiological model in which pelvic floor and abdominal muscles co-contract with the diaphragm during expiration to stabilize the trunk and regulate intra-abdominal pressure [23]. When this synergy is perturbed, the diaphragm may operate at a less favorable length or excursion, diminishing inspiratory reserve and overall vital capacity [23]. The significantly lower IC in the SUI group further supports this interpretation; while low IC can be seen in airway disease, the absence of respiratory pathology and the athletic status of our controls make a neuromuscular or synergy explanation more plausible here.
Functionally, the reduced FEV1, FVC, and FEV1/FVC ratio in SUI athletes, despite no clinical evidence of obstructive disease, likely reflects submaximal coordinated expiratory effort rather than intrinsic airflow obstruction. Prior experimental work has shown pelvic floor activity to modulate key spirometric outputs without necessarily changing static lung mechanics [14, 20]. The shorter TE in our SUI cohort complements this picture, indicating earlier cessation of forced exhalation, consistent with limited capacity to sustain high expiratory flows. Graphical analyses support the quantitative results: a blunted peak flow and a shallower early expiratory slope on flow–volume loops, together with a flatter rise in the first second and a lower plateau on time–volume curves, are typical of impaired expiratory muscle recruitment and reduced FEV1 and PEF [24].
Epidemiologically, our cohort reflects the broader pattern of elevated SUI burden in high-impact sports, where repetitive jumps, landings, and trunk bracing demand precise diaphragm–pelvic floor timing to maintain continence [18]. The present data add a mechanistic layer, showing that even in well-trained athletes, SUI is accompanied by measurable impairments on routine spirometry. Clinically, this suggests that selected spirometric markers, especially FEV1, FVC, PEF, and FEV1/FVC, can serve as accessible adjuncts for screening athletes with SUI and tracking responses to integrated respiratory–pelvic floor rehabilitation [13, 14, 20-23].
Overall, our results converge with prior evidence from laboratory, clinical, and interventional contexts: SUI is associated with altered diaphragm–pelvic floor synergy, observable as reduced inspiratory reserves and compromised expiratory flows. In athletes, where intra-abdominal pressure dynamics are repeatedly stressed, these alterations may be both more apparent and more consequential for performance and continence.
Conclusion
Semi-professional female athletes with SUI exhibit significant impairments in both inspiratory and expiratory spirometric indices compared with their healthy peers. These impairments include reduced vital capacities, diminished expiratory flows, and shortened expiratory time, suggesting dysfunctional coordination of the diaphragm, abdominal muscles, and pelvic floor. Despite their regular engagement in high-intensity sports, athletes with SUI demonstrate measurable limitations in ventilatory performance, highlighting the central role of core muscle synergy in both respiration and continence. The use of standard spirometry, particularly parameters, such as FEV1, FVC, PEF, and FEV1/FVC, may provide a valuable and accessible method for screening athletes at risk and for evaluating responses to rehabilitation programs. Targeted interventions that integrate pelvic floor and respiratory training could be essential components of management in this population.
Limitations
This cross-sectional design used limits causal inference regarding whether altered ventilatory mechanics contribute to SUI or result from it. SUI classification relied on a validated questionnaire rather than urodynamic testing, which could strengthen diagnostic precision. The sample size, while powered for primary comparisons, constrained finer subgroup analyses by sport or training load. We did not include direct measures of respiratory muscle strength or imaging of diaphragmatic excursion, which would help disentangle inspiratory versus expiratory contributions to the observed pattern. Finally, findings pertain to semi-professional athletes in selected high-impact sports and may not generalize to elite competitors, recreational athletes, or non-athletic women.
Ethical Considerations
Compliance with ethical guidelines
This study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of Iran University of Medical Sciences, Tehran, Iran (Code: IR.IUMS.REC.1403.525). As this was a retrospective study based on anonymized records, the requirement for informed consent was waived by the ethics committee.
Funding
This research did not receive any grant from funding agencies in the public, commercial, or non-profit sectors.
Authors' contributions
Conceptualization, data collection, data analysis, and drafting of the manuscript: Zahra Nazari; Study supervision, methodology, and critical revision of the manuscript: Behnoush Vassaghi Gharamaleki and Saeedeh Babazadeh.
Conflict of interest
The authors declared no conflict of interest.
References
- Rebullido TR, Gómez-Tomás C, Faigenbaum AD, Chulvi-Medrano I. The prevalence of urinary incontinence among adolescent female athletes: A systematic review. J Funct Morphol Kinesiol. 2021; 6(1):12. [DOI:10.3390/jfmk6010012] [PMID]
- Higounenc A, Carvalhais A, Vieira Á, Lopes S. Urinary incontinence in young gymnastics athletes: A scoping review. Sports. 2025; 13(9):319. [DOI:10.3390/sports13090319] [PMID]
- Kneißler E, Zentgraf K. Prevalence of urinary incontinence in female gymnasts: a systematic review. Ger J Exerc Sport Res. 2025; 55(2):159-66. [DOI:10.1007/s12662-024-01003-y]
- Salkapuram S, Keyan K, Williams H, Sivannan S, Srinivasan S, Anandakumar S. Prevalence, risk factors, and statistical analysis of urinary incontinence in a tertiary care hospital in India. Istanbul Med J. 2024; 25:286-92. [DOI:10.4274/imj.galenos.2024.09514]
- Kalani ZM, Pourmovahed ZM, Farajkhoda TP, Bagheri IM. A qualitative approach to women’s perspectives on exercise in Iran. Int J Commun Based Nurs Midwifery. 2018; 6(2):156-66. [PMID]
- Ashraf S, Kuang J, Das U, Shpenev A, Thulin E, Bicchieri C. Social beliefs and women’s role in sanitation decision making in Bihar, India: An exploratory mixed method study. Plos One. 2022; 17(1):e0262643. [DOI:10.1371/journal.pone.0262643] [PMID]
- Ron NJ, Dolbinski SC, Hodonicky EG, Middlebrook DO, Olmstead SR, Olsen SL, et al. Associations between running mechanics, functional lower extremity strength, and stress urinary incontinence in parous female runners. J Womens Pelvic Health Phys Ther. 2024; 48(3):147-53. [DOI:10.1097/JWH.0000000000000302] [PMID]
- Casey EK, Temme K. Pelvic floor muscle function and urinary incontinence in the female athlete. Phys Sportsmed. 2017; 45(4):399-407. [DOI:10.1080/00913847.2017.1372677] [PMID]
- Almousa S, Bandin Van Loon A. The prevalence of urinary incontinence in nulliparous female sportswomen: A systematic review. J Sports Sci. 2019;37(14):1663-72. [DOI:10.1080/02640414.2019.1585312] [PMID]
- Baessler K, Christmann-Schmid C, Maher C, Haya N, Crawford TJ, Brown J. Surgery for women with pelvic organ prolapse with or without stress urinary incontinence. Cochrane Database Syst Rev. 2018; 8(8):CD013108. [DOI:10.1002/14651858.CD013108] [PMID]
- Dias N, Peng Y, Khavari R, Nakib NA, Sweet RM, Timm GW, et al. Pelvic floor dynamics during high-impact athletic activities: A computational modeling study. Clin Biomech. 2017; 41:20-27. [DOI:10.1016/j.clinbiomech.2016.11.003] [PMID]
- Hay-Smith EJC, Starzec-Proserpio M, Moller B, Aldabe D, Cacciari L, Pitangui ACR, et al. Comparisons of approaches to pelvic floor muscle training for urinary incontinence in women. Cochrane Database Syst Rev. 2024; 12(12):CD009508. [DOI:10.1002/14651858.CD009508.pub2] [PMID]
- Abidi S, Ghram A, Ghroubi S, Ahmaidi S, Elleuch MH, Girard O, et al. Impact of urinary incontinence on physical function and respiratory muscle strength in incontinent women: A comparative study between urinary incontinent and apparently healthy women. J Clin Med. 2022; 11(24):7344. [DOI:10.3390/jcm11247344] [PMID]
- Park H, Han D. The effect of the correlation between the contraction of the pelvic floor muscles and diaphragmatic motion during breathing. J Phys Ther Sci. 2015; 27(7):2113-5. [DOI:10.1589/jpts.27.2113] [PMID]
- Abidi S, Ghram A, Ahmaidi S, Ben Saad H, Chlif M. Effects of inspiratory muscle training on stress urinary incontinence in north African women: A randomized controlled trial. Int Urogynecol J. 2024; 35(10):2023-31. [DOI:10.1007/s00192-024-05921-1] [PMID]
- Matsi AE, Billis E, Lampropoulou S, Xergia SA, Tsekoura M, Fousekis K. The effectiveness of pelvic floor muscle exercise with biofeedback in women with urinary incontinence: A systematic review. Appl Sci. 2023; 13(23):12743. [DOI:10.3390/app132312743]
- Mahoney K, Heidel RE, Olewinski L. Prevalence and normalization of stress urinary incontinence in female strength athletes. J Strength Cond Res. 2023; 37(9):1877-81. [DOI:10.1519/JSC.0000000000004461] [PMID]
- Pires T, Pires P, Moreira H, Viana R. Prevalence of urinary incontinence in high-impact sport athletes: A systematic review and meta-analysis. J Hum Kinet. 2020; 73:279-88. [DOI:10.2478/hukin-2020-0008] [PMID]
- Emmonds S, Heyward O, Jones B. The challenge of applying and undertaking research in female sport. Sports Med Open. 2019; 5(1):51. [DOI:10.1186/s40798-019-0224-x] [PMID]
- Park H, Hwang B, Kim Y. The impact of the pelvic floor muscles on dynamic ventilation maneuvers. J Phys Ther Sci. 2015; 27(10):3155-7. [DOI:10.1589/jpts.27.3155] [PMID]
- Hwang UJ, Lee MS, Jung SH, Ahn SH, Kwon OY. Effect of pelvic floor electrical stimulation on diaphragm excursion and rib cage movement during tidal and forceful breathing and coughing in women with stress urinary incontinence: A randomized controlled trial. Medicine. 2021; 100(1):e24158. [DOI:10.1097/MD.0000000000024158] [PMID]
- Tang Y, Guo X, Wang Y, Liu Z, Cao G, Zhou Y, et al. Rumba dance combined with breathing training as an exercise intervention in the management of stress urinary incontinence in postmenopausal women: A randomized controlled trial. Int J Environ Res Public Health. 2022; 20(1):522. [DOI:10.3390/ijerph20010522] [PMID]
- Talasz H, Kremser C, Talasz HJ, Kofler M, Rudisch A. Breathing, (s)training and the pelvic floor-a basic concept. Healthcare. 2022; 10(6):1035. [DOI:10.3390/healthcare10061035] [PMID]
- Hull JH, Ansley L, Robson-Ansley P, Parsons JP. Managing respiratory problems in athletes. Clin Med. 2012; 12(4):351-6. [DOI:10.7861/clinmedicine.12-4-351] [PMID]
Type of Study: Research |
Subject:
Physiotherapy
Received: 2025/09/20 | Accepted: 2025/12/17 | Published: 2026/03/11
Received: 2025/09/20 | Accepted: 2025/12/17 | Published: 2026/03/11








